** Progress in Earth and Planetary Science is the official journal of the Japan Geoscience Union, published in collaboration with its 51 society members.
** Progress in Earth and Planetary Science is partly financially supported by a Grant-in-Aid for Publication of Scientific Research Results to enhance dissemination of information of scientific research.
Gallery View of PEPS Articles
Research
Solid earth sciences
201511201511
An in situ experimental study of Zr4+ transport capacity of water-rich fluids in the temperature and pressure range of the deep crust and upper mantle
Mysen B
Mass transfer, Aqueous fluid, High pressure, Structure, Solubility, Spectroscopy
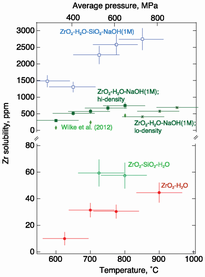
Zr concentration in fluids as a function of temperature and pressure for the systems indicated. Average pressure is that calculated from the regression line in Fig. 2. For actual pressure, see Table 1. Only two data points (at 725 and 800 °C) are shown for fluids in the ZrO2-SiO2-H2O system because at a lower temperature (and pressure) Raman signals that could be assigned to vibrations in Zr-bearing complexes in the fluid were not detected above background noise. Horizontal error bars represent calculated pressure uncertainty (±110 MPa; see text).
Throughout the Earth’s history, mass transport involved fluids. In order to address the circumstances under which Zr 4+ may have been transported in this manner, its solubility behavior in aqueous fluid with and without NaOH and SiO 2 in equilibrium with crystalline ZrO 2 was determined from 550 to 950 °C and 60 to 1200 MPa. The measurements were carried out in situ while the samples were at the temperatures and pressures of interest. In ZrO 2 -H 2 O and ZrO 2 -SiO 2 -H 2 O fluids, the Zr 4+ concentration ranges from ≤10 to ~70 ppm with increasing temperature and pressure. Addition of SiO 2 to the ZrO 2 -H 2 O system does not affect these values appreciably. In these two environments, Zr 4+ forms simple oxide complexes in the H 2 O fluid with ∆H ~ 40 kJ/mol for the solution equilibrium, ZrO 2 (solid) = ZrO 2 (fluid). The Zr 4+ concentration in aqueous fluid increases about an order of magnitude upon addition of 1 M NaOH, which reflects the formation of zirconate complexes. The principal solution mechanism is ZrO 2 + 4NaOH = Na 4 ZrO 4 + 2H 2 O with ∆H ~ 200 kJ/mol. Addition of both SiO 2 and NaOH to ZrO 2 -H 2 O enhances the Zr 4+ by an additional factor of about 5 with the formation of partially protonated alkali zircon silicate complexes in the fluid. The principal solution mechanism is 2ZrO 2 + 2NaOH + 2SiO 2 = Na 2 Zr 2 Si 2 O 9 + H 2 O with ∆H ~ 40 kJ/mol. These results, in combination with other published experimental data, imply that fluid released during high-temperature/high-pressure dehydration of hydrous mineral assemblages in the Earth’s interior under some circumstances may carry significant concentrations of Zr and probably other high field strength elements (HFSEs). This suggestion is consistent with the occurrence of Zr-rich veins in high-grade metamorphic eclogite and granulite terranes. Moreover, aqueous fluids transported from dehydrating oceanic crust into overlying mantle source rocks of partial melting also may carry high-abundance HFSE of fluids released from dehydrating slabs and transported to the source rock of partial melting in the overlying mantle wedge. These processes may have been operational in the Earth’s history within which subduction resembling that observed today was operational.