** Progress in Earth and Planetary Science is the official journal of the Japan Geoscience Union, published in collaboration with its 51 society members.
** Progress in Earth and Planetary Science is partly financially supported by a Grant-in-Aid for Publication of Scientific Research Results to enhance dissemination of information of scientific research.
Gallery View of PEPS Articles
Research
Solid earth sciences
Silicate solution, cation properties, and mass transfer by aqueous fluid in the Earth’s interior
Mysen B
Aqueous fluid, Solubility, Structure, Spectroscopy, Silicate
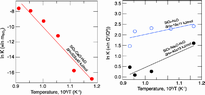
Equilibrium constants for reactions (4) (a) and (2) (b) as a function of temperature. The ∆H values are from least squares fit to Eq. (3)
Aqueous fluids in the Earth’s interior are multicomponent systems with silicate solubility and solution mechanisms strongly dependent on other dissolved components. Here, solution mechanisms that describe the interaction between dissolved silicate and other solutes were determined experimentally to 825 °C and above 1 GPa with in situ vibrational spectroscopy of aqueous fluid while these were at high temperature and pressure. The silicate content in Na-bearing, silicate-saturated aqueous fluid exceeds that in pure SiO2 at high temperature and pressure. Silicate species were of Q0 (isolated SiO4 tetrahedra) and Q1 (dimers, Si2O7) type. The temperature dependence of its equilibrium constant, K = XQ1/(XQo)2, yields enthalpies of 22 ± 12 and 51 ± 17 kJ/mol for the SiO2–H2O and Na-bearing fluids. In contrast, in Ca-bearing fluids, the solubility is more than an order of magnitude lower, and only Q0 species are present. The present data together with other published experimental information lead to the conclusion that the silicate solubility in aqueous fluids in equilibrium with mafic rocks such as amphibolite and peridotite is an order of magnitude lower than the solubility in fluids in equilibrium with felsic rocks such as andesite and rhyolite compositions (felsic gneiss) under similar temperature and pressure conditions. The silicate speciation also is more polymerized in the felsic systems. This difference is also why second critical end-points in the Earth are at lower temperature and pressure in felsic compared with mafic systems. Alkali-rich fluids formed by dehydration of felsic rocks also show enhanced high field strength element (HFSE) solubility because alkalis in such solution form oxy complexes with the HFSE cations. Fluids formed by dehydration of felsic rocks in the Earth’s interior are, therefore, more efficient transport agents of silicate materials than fluids formed by dehydration of mafic and ultramafic rocks, whether for major, minor, or trace elements.