** Progress in Earth and Planetary Science is the official journal of the Japan Geoscience Union, published in collaboration with its 51 society members.
** Progress in Earth and Planetary Science is partly financially supported by a Grant-in-Aid for Publication of Scientific Research Results to enhance dissemination of information of scientific research.
Gallery View of PEPS Articles
Research
Solid earth sciences
201508201508
Water and magmas: insights about the water solution mechanisms in alkali silicate melts from infrared, Raman, and 29 Si solid-state NMR spectroscopies
Le Losq C, Mysen B O, Cody G D
Melt, Glass, Structure, Water bonding, Water speciation, NMR spectroscopy, Raman spectroscopy, Viscosity, Magma, Fragmentation
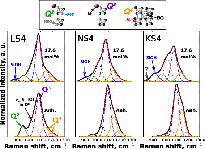
Examples of curve-fitted Raman spectra of anhydrous and 17.6 mol% H 2 O LS4 (left), NS4 (middle), and KS4 (right) glasses. Addition of water in the glasses clearly produces an increasing intensity near 900 cm −1 . It could be related to the stretching signal of Si-O-H bonds regardless of the Q n specie carrying those bonds. Signal near 850 cm −1 in the Raman spectrum of KS4 + 17.6 mol % H 2 O glass may be related to Q 1 species in the glass in negligible quantities. The various Q n species are schematically represented at the top of the figure (ionic size is not at scale). BO bridging oxygen (Si-O-Si bonds), NBO non-bridging oxygen (Si-O-M or Si-O-H bonds, with M + an alkali)
Degassing of water during the ascent of hydrous magma in a volcanic edifice produces dramatic changes in the magma density and viscosity. This can profoundly affect the dynamics of volcanic eruptions. The water exsolution history, in turn, is driven by the water solubility and solution mechanisms in the silicate melt. Previous studies pointed to dissolved water in silicate glasses and melts existing as molecules (H 2 O mol species) and hydroxyl groups, OH. These latter OH groups commonly are considered bonded to Si 4+ but may form other bonds, such as with alkali or alkaline-earth cations, for instance. Those forms of bonding influence the structure of hydrous melts in different ways and, therefore, their properties. As a result, exsolution of water from magmas may have different eruptive consequences depending on the initial bonding mechanisms of the dissolved water. However, despite their importance, the solution mechanisms of water in silicate melts are not clear. In particular, how chemical composition of melts affects water solubility and solution mechanism is not well understood. In the present experimental study, components of such information are reported via determination of how water interacts with the cationic network of alkali (Li, Na, and K) silicate quenched melts. Results from 29 Si single-pulse magic-angle spinning nuclear magnetic resonance ( 29 Si SP MAS NMR), infrared, and Raman spectroscopies show that decreasing the ionic radius of alkali metal cation in silicate melts results in decreasing fraction of water dissolved as OH groups. The nature of OH bonding also changes as the alkali ionic radius changes. Therefore, as the speciation and bonding of water controls the degree of polymerization of melts, water will have different effects on the transport properties of silicate melts depending on their chemical composition. This conclusion, in turn, may affect volcanic phenomena related to the viscous relaxation of hydrous magmas, such as for instance the fragmentation process that occurs during explosive eruptions.